The angle between the C 2 axis of the SO 2 subunit and the NCH axis is 86° and the two axes intersect at a distance of 1.38 Å above the S nucleus. Molecular Geometry: Molecules have a balanced geometric shape, the bonds. The distance between the SO 2 and HCN centers of mass is r c.m. Hydrogen cyanide (HCN) has played a central role in the production of several. The HCN molecule is roughly perpendicular to the plane of the SO 2 molecule with N lying between the SO 2 plane and the H atom. The HCN molecule has a linear molecular geometry because there is an electrical repulsion between the lone pairs of electrons in. Hydrogen cyanide is a one-carbon compound consisting of a methine group triple bonded to a nitrogen atom It has a role as a human metabolite, an Escherichia. Answered: Draw the Lewis structures of the arid. 
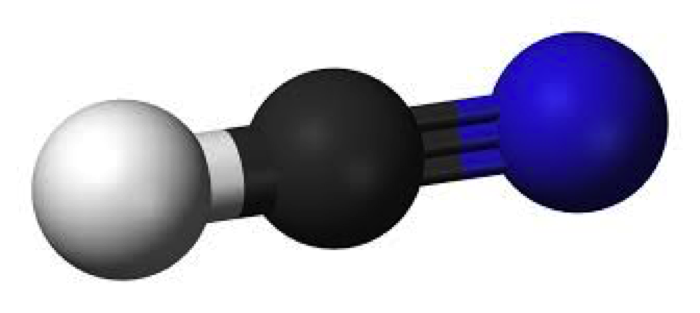
The spectroscopic constants have been interpreted in terms of a nonplanar, antihydrogen‐bonded geometry of C s symmetry with S, H, C, and N nuclei lying in the symmetry plane. molecules: Molecular Molecule Lewis Structure Molecular Geometry Polarity 1. The bond angles are 180°and two hybrid orbitals are formed for two sigma bonds formed in this molecule.The ground‐state rotational spectra of the four isotopic species ( 3 2SO 2,HC 1 4N), ( 3 2SO 2,DC 1 4N), ( 3 2SO 2,HC 1 5N), and ( 3 4SO 2,HC 1 4N) of a weakly bound dimer of sulfur dioxide and hydrogen cyanide have been observed by means of pulsed‐nozzle, Fourier‐transform microwave spectroscopy. We can say that HCN, Hydrogen Cyanide, is a linear molecule with sp hybridization to conclude this blog post. So the hybridization for HCN molecule is sp hybridization. HCN consists of three atoms: hydrogen (H), carbon (C), and nitrogen (N). This is crucial because valence electrons are the ones involved in chemical bonding. The number of sigma bonds is equal to the number of hybrid orbitals formed. Before we can start drawing the Lewis structure, we need to determine the total number of valence electrons in HCN. In HCN, there are two sigma bonds, C-H and C-N. Number of sigma bonds in the moleculeĪnother way to find out the hybridization of the molecule is to look at the number of sigma bonds formed. As central atom i carbon in HCN compound has zero lone pairs of electrons. The VSEPR theory determines the molecular geometry and form of every substance. Generally, molecules with linear molecular geometry have sp hybridization as the central atom forms bonds with two atoms only. The molecular geometry and structure of a compound are mostly determined by lone pairs, bonding electrons. There are no lone pairs of electrons on the Carbon atom, but it forms bonds with two atoms here: Nitrogen and Hydrogen.Īs the steric number is 2, referring to the table stated below, we can say that it is sp hybridized. Steric number = number of atoms attached to the central atom + number of lone pairs Hydrogen cyanide (also known as prussic acid) is a chemical compound with the formula HCN and structural formula HCN.It is a colorless, extremely poisonous, and flammable liquid that boils slightly above room temperature, at 25.6 ☌ (78.1 ☏). One can use several ways to find out the hybridization of an atom, one of them being finding the steric number for the atom. Finding out Steric Number for the molecule Chemical Lewis Structure Chemical Lewis Structure Formula Formula HCN CIF: NH3 SF2. We can find out the hybridization of the atom by using some simple and quick methods such as Steric Number and Number of sigma bonds. The carbon atom, which is also the central atom, is the one that requires hybridized orbitals to accommodate the shared electrons. The electron in its 1s orbital is shared with the Carbon atom. Hence, there is no hybridization for a Hydrogen atom as it is sharing one electron with a Carbon atom. Here the Hydrogen atom only needs one valence electron to attain a stable structure. Carbon has a single bond with a Hydrogen atom and a triple bond with a Nitrogen atom. Hydrogen Cyanide is made up of three atoms: Hydrogen, Carbon, and Nitrogen.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
